 This page contains the experiments covered in the Apologia Chemistry curriculum course, which is designed to be a first-year high school chemistry course that provides a foundation to prepare for college. The experiments listed below are those found in the textbook, so all you have to do is click the link below and watch the step by step instructional videos for each experiment.
This page contains the experiments covered in the Apologia Chemistry curriculum course, which is designed to be a first-year high school chemistry course that provides a foundation to prepare for college. The experiments listed below are those found in the textbook, so all you have to do is click the link below and watch the step by step instructional videos for each experiment.
In some cases, the experiments mentioned in the book were somewhat lacking in scope and depth (or simply didn’t work, were unsafe, or flat boring), so we’ve expanded the experiment and created something better that really covers the scientific concepts and keeps your kids excited and engaged in doing real chemistry. There’s 45 different experiments to choose from!
Most experiments have data logs and worksheets with them as well in addition to exercises to answer. If after working through the course you feel that you’d like to work through our Advanced Chemistry course, you’ll find it very comprehensive and full of instructional and lab videos not found on this page.
In order to be able to understand this text, you should have completed Algebra I. The course covers significant figures, units, classification, the mole concept, stoichiometry, thermochemistry, thermodynamics, kinetics, acids and bases, redox reactions, solutions, atomic structure, Lewis structures, molecular geometry, the gas laws, and equilibrium. You can purchase the kit to do the experiments listed in the textbook here. If you need extra chemicals, order them here. We’ve added extra experiments in addition to the ones in the book, so look them over and see which experiments you’d like to do, and then see if you can figure out way to perform the experiment with the materials and equipment you already have. Have fun!
Exploring Creation with Chemistry 2nd Edition (Click here for 3rd edition)
Step 1: Get your kids excited to learn Chemistry first by participating in this class on the basics of Chemistry.
Step 2: Work your way through the experiments listed below.
If you feel you’d like to go further, feel free to visit Unit 3, Unit 8, and Unit 15 for more chemistry experiments and key concepts. If after working through these experiments you feel that you’d like to work through our Advanced Chemistry course, you’ll find it very comprehensive and full of instructional and lab videos not found on this page.
 |
Experiment 1.1: Air Has Mass (This experiment is more fun and easier to do than the one in the textbook.) |
 |
Experiment 1.2: Air Takes Up Space |
 |
Experiment 1.3: Comparing Conversions to Measurements |
 |
Experiment 1.4: The Density of Liquids (This experiment is more fun and easier to do than the one in the textbook.) |
 |
Experiment 2.1 Calibrating your Thermometer (Make your own thermometer here, then calibrate it!) |
 |
Experiment 2.2: Measuring the Specific Heat (The Fire Balloon is one of my favorite experiments that demonstrate this scientific principle.) |
 |
Experiment 3.1: The Conservation of Mass (I don’t want students playing around with household chemicals this way, because it encourages students to mix up random stuff together without the needed safety precautions. Do this experiment instead – it’s safer and works better than the one with caustic soda.) |
 |
Experiment 3.2: Electrical Conductivity (Also try this experiment in conductivity. It takes these ideas further and connects them with physics.) |
 |
Experiment 4.1: Separating Sand and Salt This is a better experiment because it combines more of chemistry along with the filtration example. |
 |
Experiment 4.2: Physical and Chemical Change (Again, these experiments work better, are safer, and have much more fun associated with them than the ones suggested in the textbook. If you have trouble with the Instant Ice experiment, then try the Hot Icicle one instead.) |
 |
Experiment 4.3: Condensing Steam (Don’t worry about the type of can I used… just use any can you have.) |
 |
Experiment 4.4: Kinetic Theory of Matter (Here’s another convention current experiment also!) |
 |
Experiment 5.1: Measuring a Molecule (This experiment is much less complicated to do, and it works much better also!) |
 |
Experiment 6.1: Limiting Reactants (Since you’ve already done this experiment back in Chapter 1, here’s a more advanced one that allows you to change the types of reactants.) |
 |
Experiment 7.1: Electrical Charge (Also try these static experiments in addition, and if you have a Fly Stick, do this one!) |
 |
Experiment 7.2: How the Eye Detects Color (If you’d like to go further with the physics of eyes, do the Eyeballoon experiment also.) |
 |
Experiment 9.1: Covalent Bonds (There’s a teleclass that you might really like where we do static experiments.) |
 |
Experiment 9.2: Solubility of Ionic Compounds (Some kids might enjoy the Bubblology experiments also when learning about how the soap molecule is ionic at one end and covalent at the other.) |
 |
Experiment 10.1: Acids and Bases |
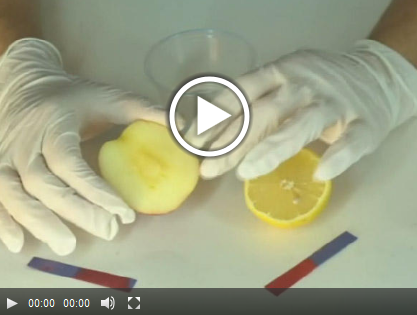 |
Experiment 10.2: Acids and Bases |
 |
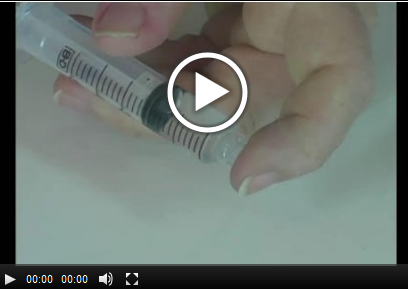
Experiment 11.1: The Effect of Temperature on Solubility (Solids) |
 |
Experiment 11.2: Effect of Temperature on Solubility (Gas) (Here’s another experiment about the triple point of water, where you can learn how to boil room temperature water!) |
 |
Experiment 11.3: Exothermic Reaction: Solute Releasing Heat (Again, I don’t want students using the chemicals stated in the book, and this one is much safer and more fun!) |
 |
Experiment 11.4: Freezing Point of Depression (I like the Instant Ice experiment because it shows you the difference between the freezing point of depression and supercooling.) |
 |
Experiment 12.1: Ideal Gas Law |
 |
Experiment 13.1: Determine the Change in Enthalpy (There’s so many cool experiments you can do with this topic, so here’s a list of them.) |
 |
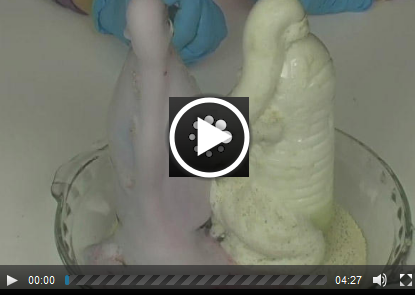
Experiment 14.1: Factors that Affect Chemical Reaction Rates (Watch the second video on the page!) |
 |
Experiment 14.2: Decomposition of Hydrogen Peroxide (Here’s the hydrogen peroxide catalyst experiment as well as another way to work with catalysts!) |
 |
Experiment 15.1: Equilibrium (I know this experiment is really different from the text, but it’s based on the same principle, and when you’re done, you’ve made a pneumatic-hydraulic earth mover you can keep!) |
 |
Experiment 15.2: Temperature and Le Chatelier’s Principle |
 |
Experiment 16.1: Invisible writing |
 Chemistry 3rd Edition
Chemistry 3rd Edition
From Apologia’s website: This revised edition offers more in-depth discussions and explanations of concepts, as well as biographical sketches of Christian scientists who have made a difference in the field while giving the glory to God. Experiments include measuring specific heat, discovering the electrical conductivity of compounds dissolved in water, measuring the width of a molecule, exploring freezing-point depression, using the ideal gas equation, recognizing the effects of catalysts, and much more.
Apologia recommends that students taking this course be proficient at an algebra math level. Topics include significant figures, units, classification, the mole concept, stoichiometry, thermochemistry, thermodynamics, kinetics, acids and basics, redox reactions, solutions, atomic structure, Lewis structures, molecular geometry, gas laws, and equilibrium.
From Aurora: If after working through the course you feel that you’d like to work through our Advanced Chemistry course, you’ll find it very comprehensive and full of instructional and lab videos not found on this page.
 |
Experiment 1.1: Air Has Mass (This experiment is more fun and easier to do than the one in the textbook.) |
 |
Experiment 1.2: Air Takes Up Space |
 |
Experiment 1.3: Comparing Conversions to Measurements |
 |
Experiment 1.4: The Density of Liquids (This experiment is more fun and easier to do than the one in the textbook.) |
 |
Experiment 2.1: The Conservation of Mass (I don’t want students playing around with household chemicals this way, because it encourages students to mix up random stuff together without the needed safety precautions. Do this experiment instead – it’s safer and works better than the one with caustic soda.) |
 |
Experiment 2.2: Electrical Conductivity (Also try this experiment in conductivity. It takes these ideas further and connects them with physics.) |
 |
Experiment 2.3: Separating Sand and Salt This is a better experiment because it combines more of chemistry along with the filtration example. |
 |
Experiment 3.1: Electrical Charge (Also try these static experiments in addition, and if you have a Fly Stick, do this one!) |
 |
Experiment 3.2: How the Eye Detects Color (If you’d like to go further with the physics of eyes, do the Eyeballoon experiment also.) |
 |
Experiment 4.1: Electron Configuration |
 |
Experiment 4.2: Valence Electrons and Lewis Dot Structures |
 |
Experiment 5.1: Covalent Bonds (There’s a teleclass that you might really like where we do static experiments.) |
 |
Experiment 5.2: Solubility of Ionic Compounds (Some kids might enjoy the Bubblology experiments also when learning about how the soap molecule is ionic at one end and covalent at the other.) |
 |
Experiment 6.2: Physical and Chemical Change (Again, these experiments work better, are safer, and have much more fun associated with them than the ones suggested in the textbook. If you have trouble with the Instant Ice experiment, then try the Hot Icicle one instead.) |
 |
Experiment 6.3: Condensing Steam (Don’t worry about the type of can I used… just use any can you have.) |
 |
Experiment 6.4: Kinetic Theory of Matter (Here’s another convention current experiment also!) |
 |
Experiment 7.1: Measuring a Molecule (This experiment is much less complicated to do, and it works much better also!) |
 |
Experiment 7.2: Mole Concept |
 |
Experiment 8.1: Limiting Reactants (Since you’ve already done this experiment back in Chapter 1, here’s a more advanced one that allows you to change the types of reactants.) |
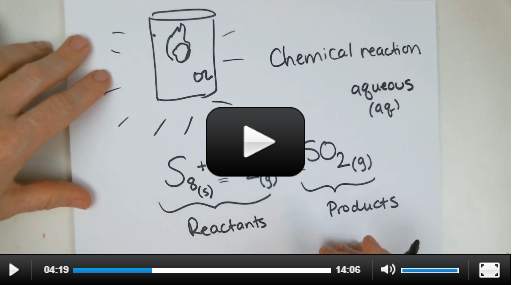 |
Experiment 8.2: Balancing Chemical Equations (find part 2 here) |
 |
Experiment 9.1: Acids and Bases |
 |
Experiment 9.2: Acids and Bases |
 |
Experiment 10.1: The Effect of Temperature on Solubility (Solids) |
 |
Experiment 10.2: Effect of Temperature on Solubility (Gas) (Here’s another experiment about the triple point of water, where you can learn how to boil room temperature water!) |
 |
Experiment 10.3: Exothermic Reaction: Solute Releasing Heat (Again, I don’t want students using the chemicals stated in the book, and this one is much safer and more fun!) |
 |
Experiment 10.4: Freezing Point of Depression (I like the Instant Ice experiment because it shows you the difference between the freezing point of depression and supercooling.) |
 |
Experiment 11.1: Ideal Gas Law |
 |
Experiment 12.1 Calibrating your Thermometer (Make your own thermometer here, then calibrate it!) |
 |
Experiment 12.2: Measuring the Specific Heat (The Fire Balloon is one of my favorite experiments that demonstrate this scientific principle.) |
 |
Experiment 13.1: Determine the Change in Enthalpy (There’s so many cool experiments you can do with this topic, so here’s a list of them.) |
 |
Experiment 14.1: Factors that Affect Chemical Reaction Rates (Watch the second video on the page!) |
 |
Experiment 14.2: Decomposition of Hydrogen Peroxide (Here’s the hydrogen peroxide catalyst experiment as well as another way to work with catalysts!) |
 |
Experiment 15.1: Equilibrium (I know this experiment is really different from the text, but it’s based on the same principle, and when you’re done, you’ve made a pneumatic-hydraulic earth mover you can keep!) |
 |
Experiment 15.2: Temperature and Le Chatelier’s Principle |
 |
Experiment 16.1: Redox and single replacement reactions (experiment: How much silver? and Salty Battery and also this Redox experiment |
 |
Experiment 16.2: Redox reactions (more experiments: fruit battery , magnesium battery, making copper ) |
